Medicine Access 101
Introduction
At Tanner Pharma, we help healthcare providers and patients obtain access to lifesaving medicines where those medicines are not available through the national healthcare system. Due to the nature of our work, we are frequently contacted by healthcare professionals, patients, and their families, friends, colleagues, or advocates who are seeking guidance and support in navigating access to necessary medical care.
In many cases, the requested treatment may be:
An investigational medicine still in clinical trials
A medicine approved in another country but not yet available locally
As a licensed distributor, Tanner Pharma can supply medicines to entities such as hospital pharmacies, retail pharmacies, local distributors, or ministries of health.
We strongly support patients advocating for their own healthcare, as this is often essential in securing the treatment they require. However, for Tanner Pharma to assist, it is vital to adhere to established processes and regulatory requirements, and to recognize that limitations within the supply chain may restrict what distributors are able to offer.
Drawing on our extensive experience, we have compiled guidance to help patients and healthcare providers navigate the complexities of accessing medicines. This article outlines how medicine access operates and explores the key factors that influence whether a particular treatment can be obtained.
Why Are Medicines Not Available in Every Country?
For a medicine to be marketed for use in humans, the health authority in a patient’s country must grant permission for general use by physicians. This permission is known as a marketing authorization (MA). Marketing authorizations are typically granted on a country-by-country basis, except in Europe where the European Medicines Agency (EMA) can grant a MA that applies to all European Member States.
It is uncommon for pharmaceutical companies to obtain marketing authorizations in every country worldwide. In fact, companies are increasingly selective about where they seek approvals, often limiting their efforts to fewer countries—particularly when it comes to treatments for rare diseases with small patient populations.
Once a MA has been granted in a country outside the United States, including by the EMA for European nations, the pharmaceutical company must subsequently engage in pricing and reimbursement (P&R) negotiations with the respective national healthcare systems. This process determines the price at which the medicine will be made available to patients and ensures agreement on how the treatment costs will be covered.
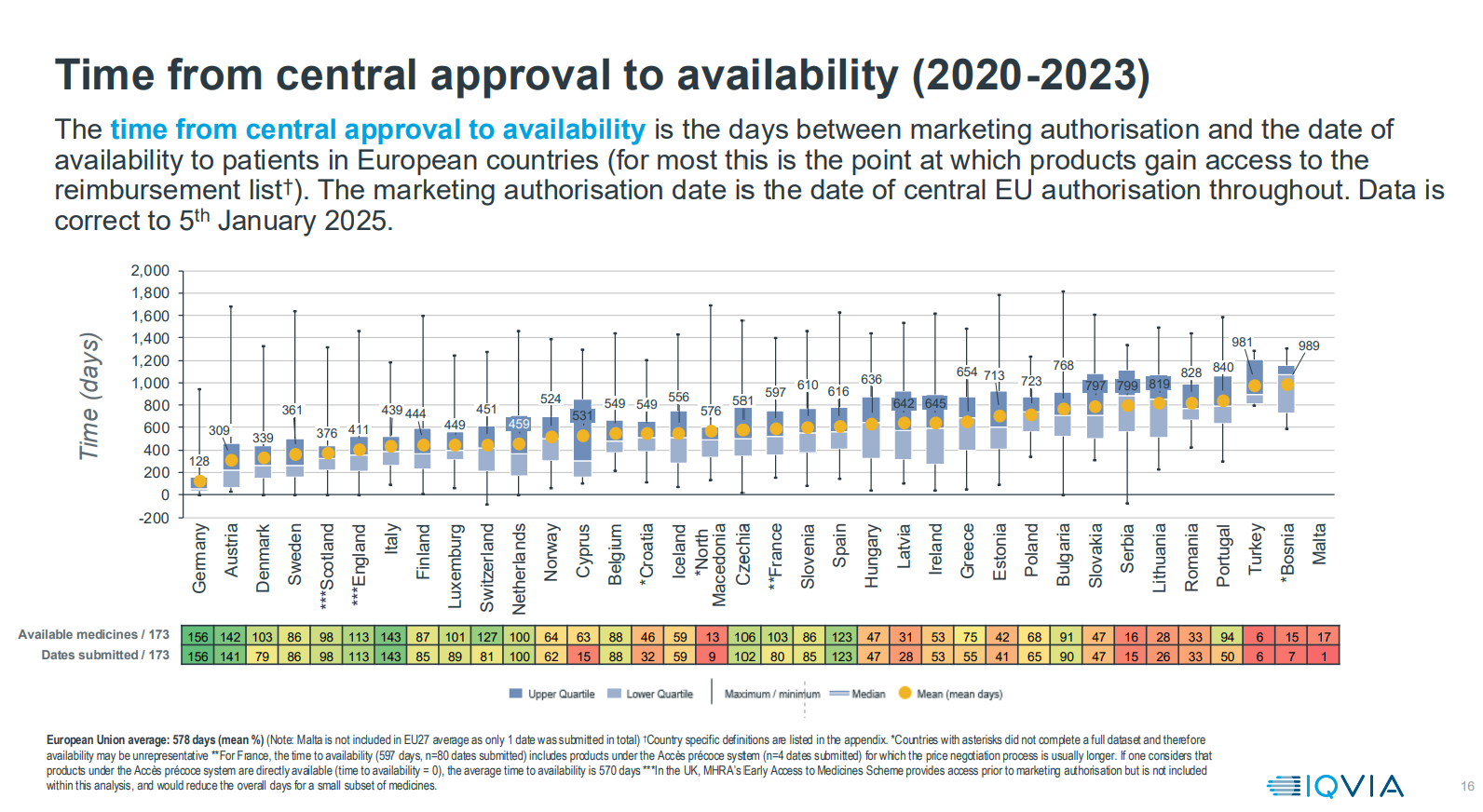
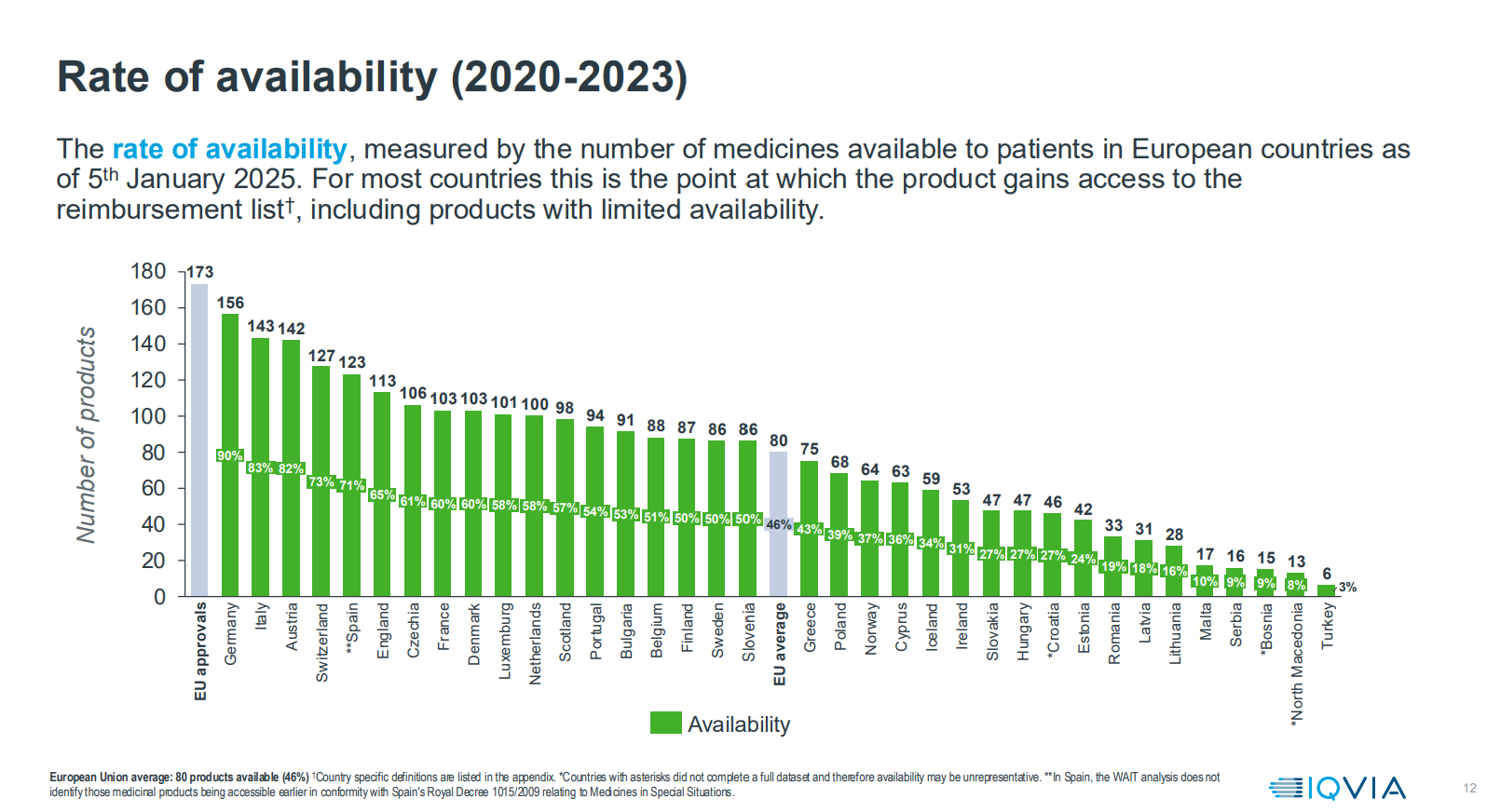
The process of making a medicine available in a country is often lengthy and complex and can sometimes lead to situations where a company decides not to make the medicine available because the price offered by the national health system is not acceptable or economically viable. Even after EMA approval is received, medicines may not be available in some European countries for several years because a company will focus resources on countries with the greatest financial opportunity first, then evaluate over time additional countries in which to consider selling a medicine.
Unfortunately, in some cases pharmaceutical companies may decide not to pursue selling a medicine in certain countries, especially when it comes to treatments for rare diseases with smaller patient populations. This is often because the limited number of patients and the anticipated pricing make it economically unviable for the company to invest in bringing the medicine to those markets.
Outside of Europe, the situation is very similar. Pharmaceutical companies will typically pursue selling a medicine in countries where there are larger patient populations and more established healthcare systems capable of paying an acceptable price for the medicine.
Examples of such countries might include:
Japan
China
Russia
Unfortunately, in most countries around the world, a company will never invest the resources needed to bring a medicine to market, and such a medicine may never be available until a company’s patent expires and a generic is available at a much lower cost.
How to Access Medicines Without Local Approval
Fortunately, most countries have regulations that allow physicians, on behalf of their patients, to obtain medicines on a case-by-case basis when no MA has been granted in their home country and the patient is not able to enroll in an existing clinical trial.
There are specific criteria that must be met to enable a physician to prescribe such a medicine and for the health system to approve such medicine to be imported into the country.
In order for a physician to prescribe a medicine that is not locally approved, and for the health system to authorize its importation into the country, there are specific criteria that must be met.
The patient cannot enroll in an ongoing clinical trial
The patient suffers from a life-threatening, long-lasting or seriously debilitating illness
There are no currently authorized therapies available to treat the disease or condition
In most cases the medicine / therapy must be undergoing clinical trials or have a marketing authorization somewhere in the world
How Does the Process Work?
Many companies offer different types of medicine access programs. These programs are often referred to as:
Expanded access
Early access
Compassionate use
Pre-approval access
Named patient supply
To obtain access to a medicine through one of these programs, a physician in the country where the patient is being treated must prescribe the medicine for treatment and participate in the process required to import the treatment into the patient’s country.
The first thing a treating physician, patient, or someone trying to help a patient should do is check the pharmaceutical company’s website see if a medicine access program is available.
Based on our experience, locating information about access programs on a company’s website can sometimes prove challenging. It may also be helpful to consult the US FDA’s clinical trial website, as these programs are frequently listed there. If you are unable to find details about a program online, we recommend reaching out directly to the company for further guidance.
When an Access Program Does Exists
If a program is available and includes the country where the patient will receive treatment, pharmaceutical companies require the treating physician to contact them directly, as the physician must determine whether the medicine is appropriate for the patient. From there, certain medical criteria needs to be met for the pharma company to approve a physician’s request; this is for the pharma company to ensure the treatment is safe for the patient and has the potential to deliver clinical benefit.
If the request is approved, the pharma company—or a third party operating the program on its behalf—will advise the physician and pharmacist on the steps needed to procure the medicine. They will also clarify whether the medicine will be provided for free, or if the patient will be required to pay either through the national health system, private insurance, out-of-pocket or through other available sources.
When an Access Program Does Not Exist
If no access program is available and the patient has a way to pay for the treatment, the physician should ask a hospital or retail pharmacist to determine whether they can locate a supplier—typically a local distributor of medicines—who can procure the medication.
If a source for obtaining the medicine is identified, in most countries it will then be necessary for the physician, pharmacist, or distributor to obtain an authorization from the local ministry of health to import the medicine into the patient’s country. The ministry of health’s website often provides information on the process to follow, though the physician or pharmacist may already be familiar with these requirements.
How Do I Pay for Such Medicines?
While some companies may offer medicines free-of-charge, many are not in a position to do so. Therefore, even if it may be possible to procure a medicine, it is important that there is a means to pay for it. Depending on the country, the national health system might pay for treatments that lack a marketing authorization, but this is not always guaranteed. If the health system will not cover the treatment, some private insurance policies may pay for it. Additionally, alternative sources such as non-profit organizations or special funds may be able to assist with payment. If none of these options are available, the patient will need to pay out-of-pocket or seek financial support through avenues like crowdsourcing.
Accessing Investigational Medicines
Many patients seek access to treatments that are still being investigated in clinical trials and do not yet have a Marketing Authorization (MA) anywhere in the world, but they are not able to do so because:
The clinical trial is fully enrolled
They do not meet the criteria to enroll in the trial
The trial is not available to patients in their country
In such cases, the only way for a patient to obtain access to an investigational medicine is if the manufacturer of the medicine is offering a program to enable access. If the manufacturer is not willing to provide the medicine for free, there are many countries where the regulations permit a company to sell an investigational medicine.